Study Design
Study population
In total, 80 people with preterm water breaking will be recruited for this study.
One group will receive vaginal probiotics and the other one will receive a vaginal placebo. The participant will be blinded and randomly assigned to the group.
Study location
This clinical study is taking place in six Quebec hospitals and two Ontario hospitals.
The three phases of the study

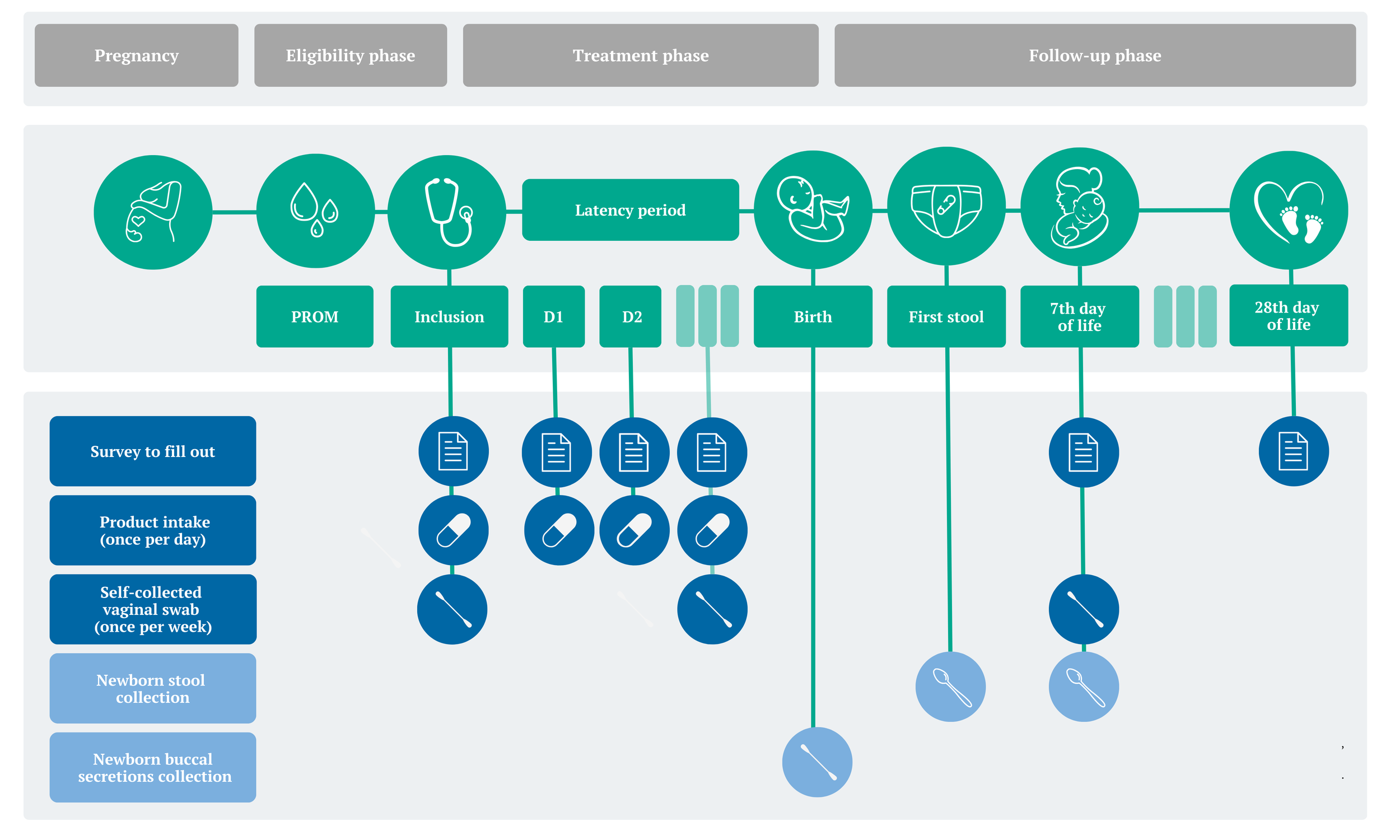
Eligibility phase
Individuals who are approached and interested in participating in the study will be evaluated by the research team to determine whether they meet the specific criteria for the study. Then, the consent and information form will be completed and signed.

Treatment phase
During this phase, participants will receive the study product on a daily basis until delivery. They will complete a logbook collecting health information and adherence to the study. Once a week, a vaginal discharge sample will be required.

Follow-up phase
After delivery, postnatal monitoring will continue for up to 28 days. This includes completing questionnaires on the baby’s health and the participants’ satisfaction with the study, as well as taking samples of the baby’s stools and the mother’s vaginal secretions for further analysis.
No travel is required during the study. All the material will be provided to ensure the participant are able to send the samples without traveling.
Study steps
Would you like to learn more?
